A unifying concept for trabecular meshwork pathology in primary open-angle glaucoma

by Ernst R. Tamm, University of Regensburg, Regensburg, Germany
IOP is generated and maintained by the aqueous humor circulation system in the anterior eye.1 Aqueous humor passes the trabecular meshwork (TM) outflow pathways which provide resistance to aqueous humor outflow and IOP builds up in response to this resistance. It is generally agreed upon that the bulk of outflow resistance in the normal eye is localized in the inner wall region of the TM outflow pathways, which comprises the juxtacanalicular connective tissue (JCT) and the inner wall endothelium of Schlemm's canal.2 Patients with primary open-angle glaucoma (POAG) typically suffer from an abnormally high outflow resistance in this region.2
There is increasing evidence that the increase in TM outflow resistance in POAG is the result of a characteristic change in the biological properties of the resident cells in the JCT, which acquire the phenotype of contractile myofibroblasts. In support of this concept are histopathological studies of human samples, cell and molecular biological experiments, and recent findings in genetically modified mouse models of POAG.
Quite similar to other resident TM cells, JCT cells constitute a mesenchymal cell population that derives from the neural crest and migrates to the chamber angle during development of the eye.3 JCT cells synthesize fibrillar extracellular matrix molecules to which they are attached via integrin-based cell-matrix contacts (Fig. 1).4 In addition, JCT cells produce non-fibrillar extracellular matrix molecules such as proteoglycans.4 There is some evidence that the quality or quantity of the JCT extracellular matrix contributes to aqueous humor outflow resistance as perfusion of anterior segment organ cultures with metalloproteinases causes an increase in outflow facility.5 Similar to the other resident TM cells, JCT cells have a distinct actomyosin cytoskeleton and show contractile properties.6,7 The actin fibers of the JCT cell cytoskeleton use the same integrin-based contacts as the extracellular fibrils and attach to their molecules from the cytoplasmic side (Fig. 1). This system allows the transmission of force and appears to be an essential requirement of the JCT to sustain its shape and function under mechanical load. Intriguingly, an increase in TM cell tone causes an increase in resistance of the TM outflow pathways,8 whereas treatment with compounds that impair integrity and/or function of the TM actomyosin system cause a substantial decrease in outflow resistance.9
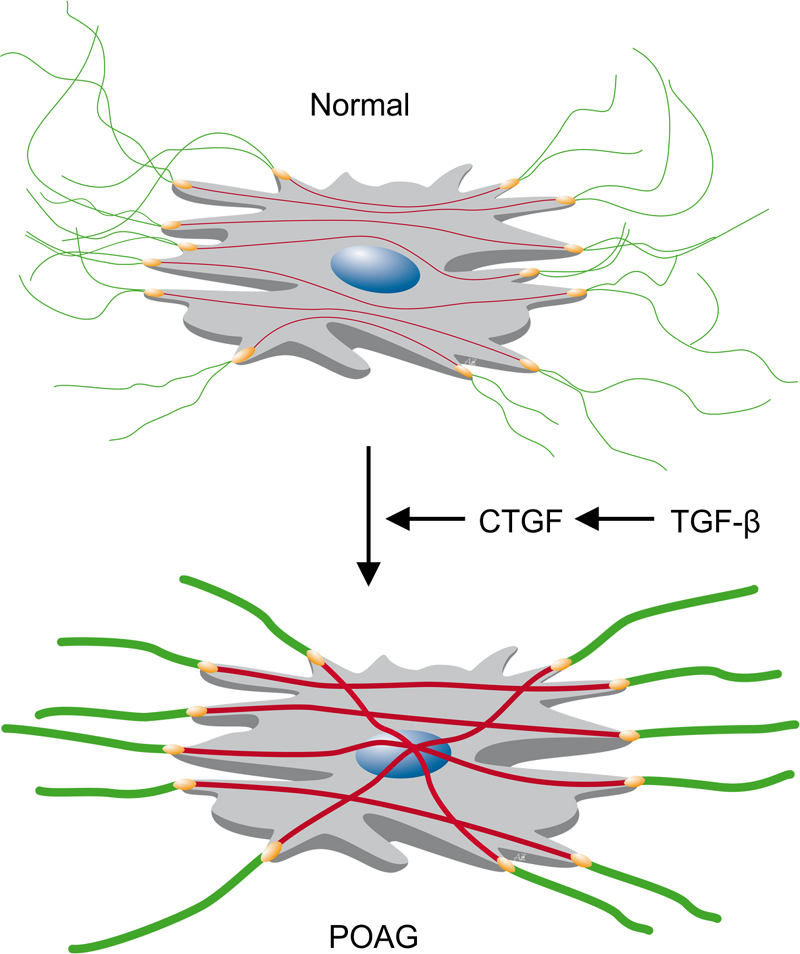
Fig. 1. Schematic drawing of the actin cytoskeleton and the fibrillar extracellular matrix of JCT cells. The force generated by the actin cytoskeleton (red) is transmitted to the fibrillar extracellular matrix (green) by integrin based cell-matrix contacts (yellow). The high activity of TGF-β signaling in POAG causes, via the downstream mediator CTGF, a switch to a myofibroblast-like phenotype including an augmentation of the actin cytoskeleton and its directly associated extracellular fibrillar matrix. Overall, the changes cause an increase in TM rigidity and aqueous humor outflow resistance.20
In POAG, the extracellular matrix of the JCT shows characteristic histopathological changes that include the increase in extracellular fibrillar material which is associated with the JCT elastic fibers and has been termed plaque-material.10 While the amount of plaque material correlates with the axonal loss in the optic nerve, highly conserved signaling molecule that consists of three isoforms (TGF-β1, -β2, and -β3) in mammals.12
There is some evidence that the quality or quantity of the JCT extracellular matrix contributes to aqueous humor outflow resistance as perfusion of anterior segment organ cultures with metalloproteinases causes an increase in outflow facility.
Quite noteworthy, treatment of TM cells with TGF-β1 or TGF-β2 not only induces the production of TM cell extracellular matrix synthesis, but also increases the contractile properties of the TM actin cytoskeleton.12 In the eye, TGF-β2 is found in high amounts in the aqueous humor and vitreous, while TM cells are capable of synthesizing both TGF-β1 and TGF-β2.12 Most patients with POAG show increased amounts of the activated form of TGF-β2 in their aqueous humor.12 It is tempting to speculate that the increase in TGF-β activity in the aqueous humor of patients with POAG directly causes the accumulation of the JCT extracellular matrix and increases the contractility of JCT cells, and that both factors cause or contribute to the increase in TM outflow resistance in eyes with POAG.
It is tempting to speculate that the increase in TGF-β activity in the aqueous humor of patients with POAG directly causes the accumulation of the JCT extracellular matrix and increases the contractility of JCT cells, and that both factors cause or contribute to the increase in TM outflow resistance in eyes with POAG.
Direct support for this hypothesis comes from recent studies in transgenic mice that develop POAG.13 In the eye, TGF-β signaling is embedded in a tightly controlled homeostatic signaling network that consists of molecules which promote or inhibit the activity of TGF-β signaling.12,14 A critical downstream mediator of TGF-β signaling is the molecule connective tissue growth factor (CTGF) which is highly expressed in normal TM cells.15 CTGF is critically required both for mediating the TGF-β-induced effects on extracellular matrix synthesis and for increasing the contractile properties of mesenchymal cells including that of the TM.13,16 Transgenic mice that overexpress CTGF in their eyes show an increase in IOP that correlates with an axonal loss in their optic nerve.13 The chamber angle of CTGF-overexpressing mice is wide open, a finding which justifies the assumption that the mice constitute an animal model with all essential characteristics of POAG. Quite intriguingly, the TM of CTGF-overexpressing mice with POAG shows an accumulation of fibrillar extracellular matrix along with an increase in actin-based contractility. Treatment of CTGF-overexpressing mice with a Rhokinase inhibitor that disrupts their TM actin fibers reduces within hours their high IOP to levels seen in normal animals.13 Overall, the effects of CTGF on IOP appear to be mediated by a modification of the TM actin cytoskeleton. The observations lead to the assumption that the high activity of TGF-β signaling in POAG causes, via the downstream mediator CTGF, a change in the phenotype of JCT cells. The cells switch to a myofibroblast-like phenotype, a scenario which strengthens simultaneously both their actin cytoskeleton and their directly associated extracellular matrix fibrils (Fig. 1). Overall, the changes cause an increase in TM rigidity and aqueous humor outflow resistance.
Impaired TGF-β signaling might be the common causative theme in POAG.
Quite intriguingly, such a scenario might not only happen in the TM of patients with POAG. There is evidence that CTGF causes comparable effects in optic nerve astrocytes17 and lamina cribrosa cells.18 Moreover, a change to a myofibroblasts-like phenotype has been also observed in scleral fibroblasts along the peripapillary region of eyes with experimental glaucoma.19 Impaired TGF-β signaling might be the common causative theme in POAG.
References
- Tamm ER, Toris CB, Crowston JG, et al. Basic science of intraocular pressure. In: Weinreb RN, Brandt JD, Garway-Heath D, Medeiros F (eds.), Intraocular pressure Reports and consensus statements of the 4th global AIGS consensus meeting on intraocular pressure. Amsterdam: Kugler Publications 2007; pp. 1-14.
- Johnson M. What controls aqueous humour outflow resistance? Exp Eye Res 2006;82:545-557.
- Cvekl A, Tamm ER. Anterior eye development and ocular mesenchyme: new insights from mouse models and human diseases. Bioessays 2004;26:374-386.
- Acott TS, Kelley MJ. Extracellular matrix in the trabecular meshwork. Exp Eye Res 2008;86:543-561.
- Bradley JM, Vranka J , Colvis CM, et al. Effect of matrix metalloproteinases activity on outflow in perfused human organ culture. Invest Ophthalmol Vis Sci 1998;39:2649-2658.
- Tian B, Gabelt BT, Geiger B, Kaufman PL. The role of the actomyosin system in regulating trabecular fluid outflow. Exp Eye Res 2009;88:713-717.
- Tian B, Geiger B, Epstein DL, Kaufman PL. Cytoskeletal involvement in the regulation of aqueous humor outflow. Invest Ophthalmol Vis Sci 2000;41:619-623.
- Wiederholt M, Thieme H, Stumpff F. The regulation of trabecular meshwork and ciliary muscle contractility. Prog Retin Eye Res 2000;19:271-295.
- Rao VP, Epstein DL. Rho GTPase/Rho kinase inhibition as a novel target for the treatment of glaucoma. BioDrugs 2007;21:167-177.
- Tektas OY, Lütjen-Drecoll E. Structural changes of the trabecular meshwork in different kinds of glaucoma. Exp Eye Res 2009;88:769-775.
- Gottanka J, Johnson DH, Martus P, Lutjen-Drecoll E. Severity of optic nerve damage in eyes with POAG is correlated with changes in the trabecular meshwork. J Glaucoma 1997;6:123-132.
- Fuchshofer R, Tamm ER . The role of TGF-beta in the pathogenesis of primary open-angle glaucoma. Cell Tissue Res 2012;347:279-290.
- Junglas B, Kuespert S, Seleem AA, et al. Connective tissue growth factor causes glaucoma by modifying the actin cytoskeleton of the trabecular meshwork. Am J Pathol 2012;180:2386-2403.
- Fuchshofer R, Tamm ER . Modulation of extracellular matrix turnover in the trabecular meshwork. Exp Eye Res 2009;88:683-688.
- Junglas B, Yu AH, Welge-Lussen U, Tamm ER, Fuchshofer R. Connective tissue growth factor induces extracellular matrix deposition in human trabecular meshwork cells. Exp Eye Res 2009;88:1065-1075.
- Fuchshofer R, Ullmann S, Zeilbeck LF, Baumann M, Junglas B, Tamm ER. Connective tissue growth factor modulates podocyte actin cytoskeleton and extracellular matrix synthesis and is induced in podocytes upon injury. Histochem Cell Biol 2011;136:301-319.
- Fuchshofer R. The pathogenic role of transforming growth factor-beta2 in glaucomatous damage to the optic nerve head. Exp Eye Res 2011;93:165-169.
- Wallace DM, Clark AF, Lipson KE, Andrews D, Crean JK, O'Brien CJ. Anticonnective tissue growth factor antibody treatment reduces extracellular matrix production in trabecular meshwork and lamina cribrosa cells. Invest Ophthalmol Vis Sci 2013;54:7836-7848.
- Quigley HA, Cone FE. Development of diagnostic and treatment strategies for glaucoma through understanding and modification of scleral and lamina cribrosa connective tissue. Cell Tissue Res 2013;353:231-244.
- Tamm ER. Functional morphology of the outflow pathways of aqueous humor and their changes in open angle glaucoma. Ophthalmologe 2013;110:1026-1035.


